
Terveystalo Clinical Research
Terveystalo Clinical Research (TCR) is the largest site management unit in Finland. We have obtained strong knowledge and experience of clinical trials since 1992.
TCR offers high-quality clinical research services in compliance with ICH-GCP. We currently manage 6 clinical trial sites strategically located across the country, offering the capacity of approximately 360 clinics in about 100 localities with 14,800 healthcare professionals in Terveystalo. There are many national opinion leaders amongst them in several therapeutic areas as well as trained Coordinating Investigators. We have both GCP-compliant investigators and study coordinators.
Apart from clinical drug trials, we can also conduct trials on nutraceuticals and biomaterial studies.
High experience at our client's service
TCR provides easy access to successful trials:
- free feasibility assessment
- fast site and investigator selection
- fast trial subject prescreening using our huge EMR database
- experienced and GCP-compliant Investigators and Coordinators
- centralised budget and contract negotiations
- opinion leaders in several therapeutic areas
- trained National Coordinating Investigators
- multiple recruitment sources
- Electronic Medical Record (EMR) system in use by all physicians in Terveystalo
- laboratory and imaging services available
- centralised invoicing
| Our values: | Our mission: | Our vision: |
|---|---|---|
| Competence - Caring - Continuous improvement | Clinical trial services in the best interests of human health | TCR – The leading Site Management Unit in Finland |
Contact information:

Riina-Riitta Helminen
Director, Terveystalo Clinical Research
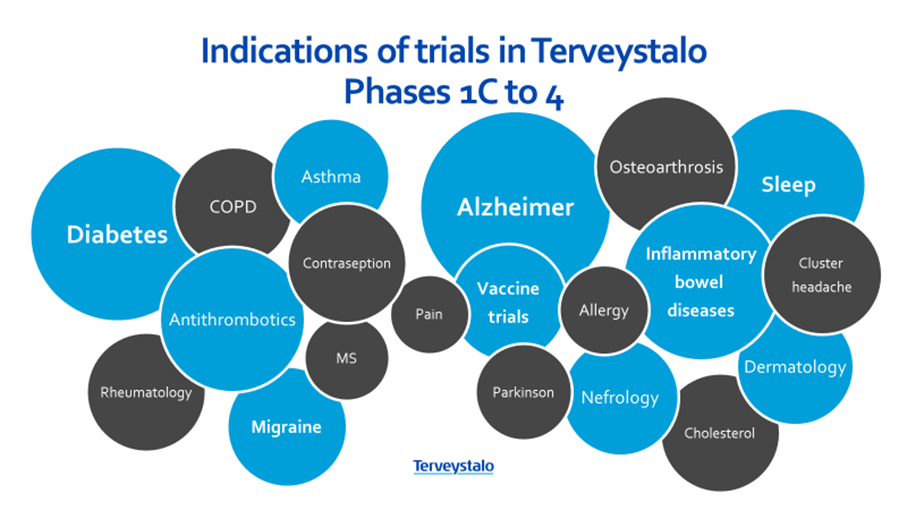
- More than 30 trials ongoing
- Over 500 individual trial subjects per year
- Around 3,000 trial related visits per year
- Largest laboratory network in Finland
- The largest site management unit in Finland
- Centralized contract negotiation, budgeting and invoicing
- Search for investigators is free of charge

Terveystalo operates in over 100 localities across Finland
The largest healthcare service company in Finland.
We develop Finnish healthcare by measuring clinical and operational quality and the quality of customer experiences and by introducing new electronic services to the customers.
360 clinics
18 hospitals
31 dental clinics
740,000 people receiving occupational healthcare services
1 200,000 individual customers
over 14,000 healthcare professionals
15% appointments with physicians in Finland